The Food and Drug Administration (FDA) rolled out a new platform, backed by artificial intelligence, that will streamline publicly accessible reporting of negative or unexpected health effects linked to medicines, vaccines, cosmetics, animal food and other consumer products. The FDA Adverse Event Monitoring System (AEMS) began operation Tuesday and will consolidate outdated systems used to
All posts in "Politics"
The Food and Drug Administration (FDA) rolled out a new platform, backed by artificial intelligence, that will streamline publicly accessible reporting of negative or unexpected health effects linked to medicines, vaccines, cosmetics, animal food and other consumer products. The FDA Adverse Event Monitoring System (AEMS) began operation Tuesday and will consolidate outdated systems used to
650
March 20, 2026House Majority Leader Steve Scalise, R-La., is hammering Democrats over the partial government shutdown as its effects begin to hit millions of travelers at airports across the country. ‘This is expected to be one of the busiest spring travel seasons on record. Over 171 million travelers are estimated to fly in the coming weeks, and
House Majority Leader Steve Scalise, R-La., is hammering Democrats over the partial government shutdown as its effects begin to hit millions of travelers at airports across the country. ‘This is expected to be one of the busiest spring travel seasons on record. Over 171 million travelers are estimated to fly in the coming weeks, and
653
March 20, 2026Republicans sharply criticized former President Joe Biden over rising prices at the gas pump, but a spike in energy prices amid the U.S.-Israeli conflict in Iran threatens to scramble the party’s affordability messaging. The Iran conflict has led to a surge in gas prices for Americans, leading to an average 50 cents a gallon increase
Republicans sharply criticized former President Joe Biden over rising prices at the gas pump, but a spike in energy prices amid the U.S.-Israeli conflict in Iran threatens to scramble the party’s affordability messaging. The Iran conflict has led to a surge in gas prices for Americans, leading to an average 50 cents a gallon increase
589
March 20, 2026A suspected retaliatory drone attack by pro-Iranian militias struck a major U.S. diplomatic facility in Baghdad on Tuesday, according to The Washington Post. The newspaper said the strike hit the Baghdad Diplomatic Support Center, and no injuries were immediately reported. Six drones were launched toward the compound, five of which were shot down. The Post,
A suspected retaliatory drone attack by pro-Iranian militias struck a major U.S. diplomatic facility in Baghdad on Tuesday, according to The Washington Post. The newspaper said the strike hit the Baghdad Diplomatic Support Center, and no injuries were immediately reported. Six drones were launched toward the compound, five of which were shot down. The Post,
1K
March 12, 2026President Donald Trump took a bow Tuesday night for his 5-0 record for his endorsed candidates in the Republican elections held in Mississippi and Georgia. ‘March 10th election update: 5 wins, 0 losses,’ an election night image posted to Truth Social blared. ‘President Trump endorsements 100%.’ The image hailed a 4-0 record in Mississippi (Sen.
President Donald Trump took a bow Tuesday night for his 5-0 record for his endorsed candidates in the Republican elections held in Mississippi and Georgia. ‘March 10th election update: 5 wins, 0 losses,’ an election night image posted to Truth Social blared. ‘President Trump endorsements 100%.’ The image hailed a 4-0 record in Mississippi (Sen.
725
March 12, 2026About 60% of Texas Republicans voted last Tuesday to end John Cornyn’s career in the Senate, but it wasn’t really Cornyn they were rejecting. It was the feckless, do-nothing GOP Senate leadership that makes ‘Waiting for Godot’ look like a ‘Fast and Furious.’ Texas Attorney General Ken Paxton wound up in a virtual tie with
About 60% of Texas Republicans voted last Tuesday to end John Cornyn’s career in the Senate, but it wasn’t really Cornyn they were rejecting. It was the feckless, do-nothing GOP Senate leadership that makes ‘Waiting for Godot’ look like a ‘Fast and Furious.’ Texas Attorney General Ken Paxton wound up in a virtual tie with
642
March 12, 2026A group of House Republicans is urging the Trump administration to choke off Russia’s profits from one of its largest energy companies as global oil prices spike. It comes as the U.S. and Israel’s conflict with Iran, one of Russia’s closest allies and another major energy producer, is threatening to spiral the market out of
A group of House Republicans is urging the Trump administration to choke off Russia’s profits from one of its largest energy companies as global oil prices spike. It comes as the U.S. and Israel’s conflict with Iran, one of Russia’s closest allies and another major energy producer, is threatening to spiral the market out of
588
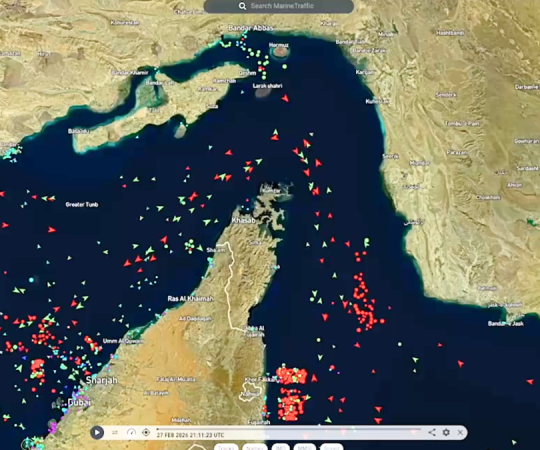
March 12, 2026U.S. forces destroyed 16 Iranian mine-laying vessels near the Strait of Hormuz Tuesday, U.S. Central Command said, in what officials described as a move to prevent Iran from disrupting one of the world’s most critical maritime choke points. The strikes come as oil traffic through the strait remains at a near standstill, threatening a corridor
U.S. forces destroyed 16 Iranian mine-laying vessels near the Strait of Hormuz Tuesday, U.S. Central Command said, in what officials described as a move to prevent Iran from disrupting one of the world’s most critical maritime choke points. The strikes come as oil traffic through the strait remains at a near standstill, threatening a corridor
3.3K
December 23, 2025China has reportedly loaded more than 100 intercontinental ballistic missiles into three newly constructed silo fields near its border with Mongolia and shows little interest in arms control talks, according to a draft Pentagon report seen by Reuters. The assessment underscores Beijing’s accelerating military buildup, with the report saying China is expanding and modernizing its
China has reportedly loaded more than 100 intercontinental ballistic missiles into three newly constructed silo fields near its border with Mongolia and shows little interest in arms control talks, according to a draft Pentagon report seen by Reuters. The assessment underscores Beijing’s accelerating military buildup, with the report saying China is expanding and modernizing its
1.3K
December 23, 2025The Social Security Administration’s (SSA) internal watchdog has confirmed that the agency’s publicly reported phone service data was accurate and that performance improved during fiscal year 2025, according to a new audit completed after Sen. Elizabeth Warren, D-Mass., questioned whether the figures could be trusted. The Office of the Inspector General (OIG) reviewed the SSA’s
The Social Security Administration’s (SSA) internal watchdog has confirmed that the agency’s publicly reported phone service data was accurate and that performance improved during fiscal year 2025, according to a new audit completed after Sen. Elizabeth Warren, D-Mass., questioned whether the figures could be trusted. The Office of the Inspector General (OIG) reviewed the SSA’s
Emily
Emily is a passionate financial writer and market enthusiast with over a decade of experience in personal finance, stock market trends, and long-term investing strategies. With a background in economics and a love for simplifying complex financial topics, she helps readers make smarter money decisions with confidence. When she’s not analyzing charts or writing about the latest market moves, Emily enjoys hiking, strong coffee, and reading biographies of great investors.
Recent Posts
- Tether Commits $127.5M to Drift Protocol Post Hack: How Will Affected Users Get Paid?
- Crypto News: Why Pepeto Could Beat XRP Price Gains as Rakuten Opens XRP to 44 Million Users
- Cardano (ADA) Price Prediction 2026, 2027 – 2030: Will ADA Price Hit $2?
- Bitcoin Sentiment Signals Hint at a Classic Contrarian Setup
- Dogecoin Breakout Confirmed After Third Attempt Flips Resistance